How vaccines work
Vaccines work by teaching the immune system how to fight off a disease in case it ever comes into contact with it. This greatly reduces the risk of becoming seriously ill or spreading a disease to others. Vaccines can protect against one or multiple diseases. Sometimes, multiple vaccines may be given at once to protect against several diseases.
Vaccines can protect against either one or multiple diseases. Sometimes, multiple vaccines may be given at once to protect against several diseases.
Most vaccines contain a weakened or an inactivated form of a virus or bacterium, or a small part of it, called an antigen.
When a person gets vaccinated, their immune system recognises the antigen as foreign. This activates immune cells to produce antibodies and create a memory of the virus or bacterium.
Later, if the person comes into contact with the actual virus or bacterium, their immune system will remember it, and then produce the right antibodies and activate the right immune cells quickly, to kill the virus or bacterium. This protects the person from the disease.
In contrast, people who become immune by getting the actual disease can spread it to others and put themselves at risk of serious complications from the disease.
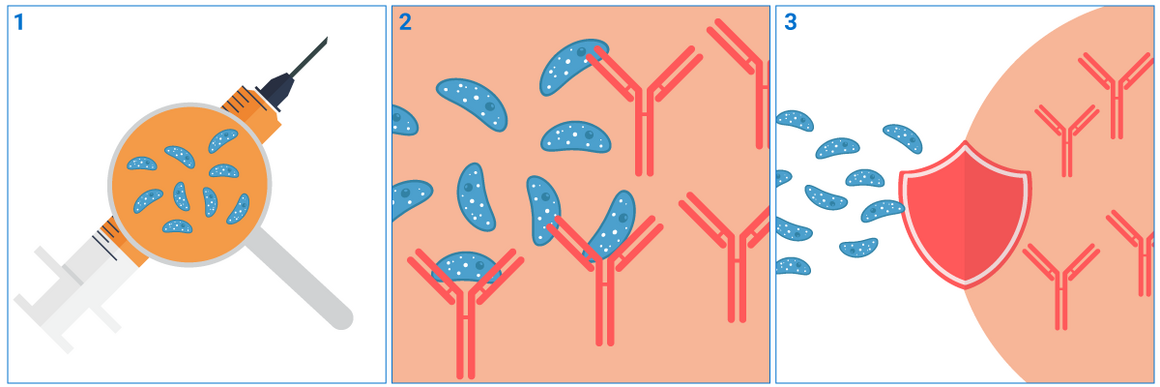
Protection
Different vaccines bring about different levels of protection. The duration of protection varies depending on the disease. Some vaccines can only protect against a disease for a short period and may need booster doses; for others, immunity can last a lifetime.
Vaccination not only protects people who have received a vaccine. It also indirectly protects unvaccinated people in the community, by decreasing the risk of exposure to infection. This is known as community immunity (also called herd immunity).
Components
In addition to one or more antigens, other components can also be added to help keep the vaccine stable and effective. Regulators make sure that all these components are safe.
These components include:
- stabilisers: to keep vaccine components stable;
- adjuvants: these improve the immune response to the vaccine by making the response stronger, faster and more sustained over time – an example of which is aluminium;
- excipients: these are inactive ingredients, like water, or sodium chloride (salt), as well as preservatives or stabilisers that help the vaccine remain unchanged during storage, keeping it active.
In some vaccines, there may also be trace amounts of other substances used in the manufacturing process, such as ovalbumin (a protein found in eggs) or neomycin (an antibiotic). If these substances might cause a reaction in sensitive or allergic individuals, their presence is declared in the information given to healthcare workers and patients.
Types of vaccines
Vaccines based on proteins contain small pieces of a virus or bacteria that are not dangerous but help the immune system recognise and fight a real infection. The flu, tetanus and whooping cough vaccines are examples of this type of vaccine and have been in use for many years.
These proteins, made in a laboratory, only stimulate the immune system and do not cause an infection or disease.
Protein-based vaccines often contain substances called adjuvants. These strengthen the immune system’s response to the vaccine and increase protection.
mRNA and viral-vector vaccines contain instructions for the human cells that tell them how to make an antigen protein. These instructions come in one of two forms, either as a molecule called messenger ribonucleic acid or mRNA, or a harmless virus that carries genetic information.
When a person receives one of these types of vaccines, their cells follow these instructions and then produce the antigen protein which the immune system recognises as foreign, activating immune cells and creating antibodies.
The first four vaccines against COVID-19 authorised in the EU were mRNA or viral-vector vaccines.
Approval of vaccines in the EU
Find out how authorities in Europe test to make sure vaccines are safe and effective before they are approved for use.
Benefits of vaccination
How do vaccines protect us and stop the spread of disease? Find out their benefits for individuals and the community.
COVID-19 medicines
European Medicines Agency - Safety of COVID-19 vaccines
